Berberine is a quaternary benzylisoquinoline plant alkaloid that is transforming the management of SIBO. Chemically identified as 5,6-dihydro-9,10-dimethoxybenzo[g]-1,3-benzodioxolo[5,6-a]quinolizinium, this highly bioactive compound is primarily synthesised in the roots, rhizomes and bulbs of medicinal plants belonging to the Berberidaceae, Ranunculaceae and Papaveraceae families. Among the most documented botanical sources: Coptis chinensis (Goldenthread), Phellodendron amurense (Amur cork tree), Berberis vulgaris (Barberry), Berberis aquifolium (Oregon grape) and Berberis aristata (Indian Barberry).
Historically used in traditional Ayurvedic and Chinese pharmacopoeias to treat bacterial gastroenteritis, secretory diarrhoea, typhoid fever and inflammatory conditions, berberine is now the subject of intensive pharmacological research. The most recent data — notably the BRIEF-SIBO randomised trial of 2024 and a network meta-analysis of 30 RCTs — position it as the top-ranked intervention for SIBO eradication, ahead of rifaximin monotherapy.
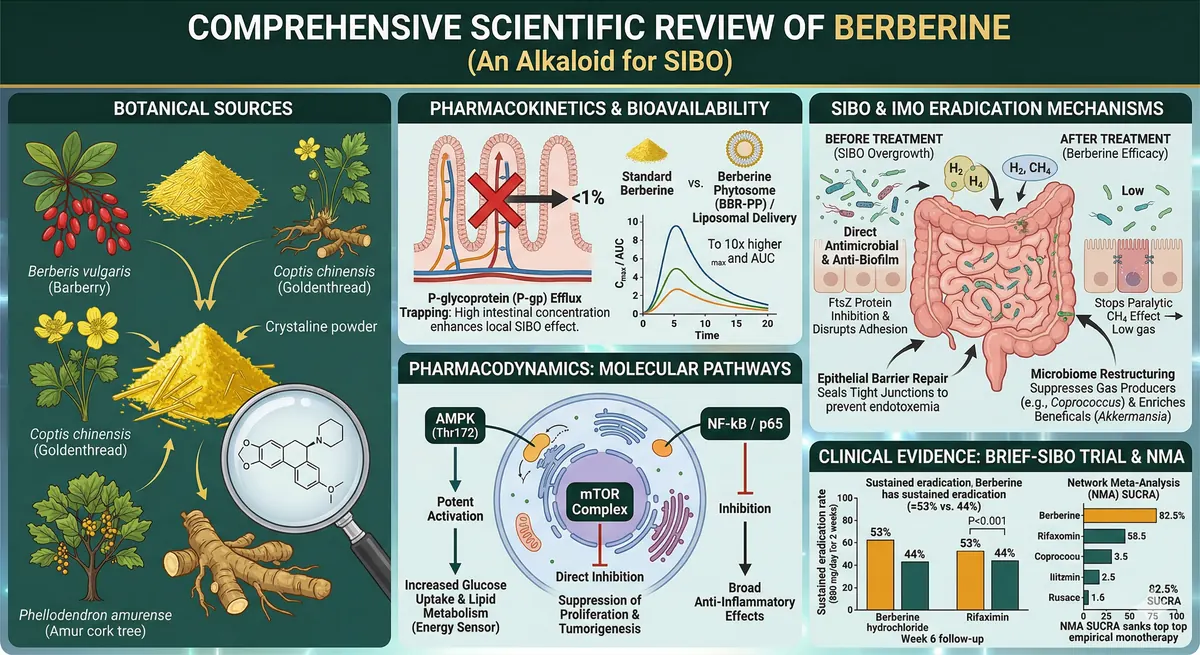
1. Pharmacokinetics, Bioavailability and Formulation Innovations
Despite a remarkable pharmacological profile, the systemic clinical utility of unformulated berberine is severely limited by an oral bioavailability below 1%. This constraint is primarily explained by extensive first-pass hepatic metabolism and active efflux via P-glycoprotein (P-gp).
1.1 The P-glycoprotein trap: a disguised advantage
P-gp is an ABC (ATP-binding cassette) efflux transporter, highly expressed on the apical membrane of enterocytes. When berberine passively diffuses into the enterocyte, P-gp actively pumps it back into the intestinal lumen — trapping virtually the entire administered dose within the gastrointestinal tract.
Key insight: This efflux mechanism, traditionally considered a limitation, paradoxically constitutes a major therapeutic advantage for localised gastrointestinal conditions such as SIBO. Berberine achieves high intraluminal concentrations precisely where the pathological bacterial overgrowth is located.
1.2 Formulation strategies for systemic exposure
For systemic applications (metabolic syndrome, type 2 diabetes), advanced formulation technologies have been developed:
- Co-administration of P-gp and CYP inhibitors: silymarin, pluronic P85, tween 80 — increase plasma exposure
- Lipid-based systems: nanoencapsulation, salt formation, mucoadhesive microparticles
- Berberine phytosome (BBR-PP): phospholipid complex with pea proteins and procyanidolic oligomers — bioavailability up to 10 times higher
| Formulation | Cmax (ng/mL) | AUC (ng·h/mL) | Improvement |
|---|---|---|---|
| Standard berberine | 1.67 ± 0.41 | 13.4 ± 1.97 | Reference |
| Liposomal berberine (LMB) | 15.8 ± 2.6 | 61.1 ± 11.2 | ×9.5 (Cmax), ×4.6 (AUC) |
2. Pharmacodynamics: Molecular Pathways
Berberine acts on a remarkably dense network of molecular targets, which explains its exceptionally broad therapeutic spectrum.
2.1 AMPK activation
Berberine potently activates AMP-activated protein kinase (AMPK) at the Thr172 residue — the master regulator of cellular energy homeostasis. This activation is LKB1-independent. Downstream: inhibition of acetyl-CoA carboxylase (ACC), suppression of fatty acid synthesis, enhanced glucose uptake, improved insulin sensitivity and thermogenesis stimulation.
2.2 mTOR inhibition
Via the AMPK pathway, berberine inhibits mTOR (mammalian target of rapamycin), suppressing cell proliferation and tumourigenesis. It induces p53 phosphorylation and caspase-3 cleavage — triggering apoptosis in malignant cells (demonstrated in colorectal lines HCT116, SW480, LOVO).
2.3 NF-κB suppression
Berberine inhibits NF-κB/p65 phosphorylation via an AMPK-independent mechanism. It downregulates COX-2 and modulates the TLR4/MyD88/NF-κB pathway, conferring broad-spectrum anti-inflammatory effects particularly relevant in the context of chronic intestinal inflammation associated with SIBO.
2.4 Additional targets
- KCNH-6: regulation of insulin secretion
- SIRT-1: cellular longevity and lipid metabolism
- HIF-1α and VEGF: angiogenesis
- PI3K/Akt: cell survival
- JAK-2: cytokine signalling
- Ca2+ channels: cellular excitability
- Endoplasmic reticulum stress markers
3. Systemic Clinical Applications
3.1 Metabolic syndrome and type 2 diabetes
Berberine promotes insulin secretion, alleviates insulin resistance, inhibits hepatic gluconeogenesis and blocks α-amylase and α-glucosidase. Clinical studies report significant reductions in fasting blood glucose, triglycerides, waist circumference and BMI. Doses above 1 g/day for more than 8 weeks show efficacy comparable to metformin in pilot studies.
3.2 Cardiovascular protection
Berberine modulates lipid metabolism, prevents diabetic cardiomyopathy, halts cardiac fibrosis and restores endothelial function via the SIRT-1 and PI3K/Akt pathways. Clinical data support its efficacy in ischaemic heart disease, stroke, arrhythmias and aortic aneurysms.
4. SIBO: Pathophysiology and the Migrating Motor Complex
4.1 Definition and diagnosis
Under normal physiological conditions, the small intestine maintains relative sterility with bacterial concentrations below 105 CFU/mL. SIBO is diagnosed when bacterial concentrations exceed this threshold in proximal jejunal fluid, or by breath test: ≥20 ppm hydrogen rise or ≥10 ppm absolute methane within 90 minutes (North American Consensus).
4.2 Clinical presentation
SIBO symptoms include: intense abdominal pain, severe distension, refractory bloating, chronic cramping, flatulence, eructation, irregular bowel movements, malabsorption, vitamin B12 deficiency and weight loss.
4.3 The migrating motor complex (MMC): guardian of the small intestine
The MMC is the inter-digestive cleansing mechanism of the small intestine. It comprises three phases:
| Phase | Duration | Characteristics |
|---|---|---|
| Phase I | 55% of cycle | Complete quiescence |
| Phase II | 41% of cycle | Irregular low-amplitude contractions |
| Phase III | 4% (5–10 min) | Powerful peristaltic wave from stomach to terminal ileum — sweeps bacteria into the colon |
Phase III, mediated by motilin and vagal innervation, is the essential guardian of small intestinal sterility. Its dysfunction is the primary cause of SIBO.
4.4 Causes of MMC failure
- Vagus nerve damage: abdominal surgery, diabetic neuropathy
- Post-surgical adhesions
- Chronic stress: sustained vagal inhibition
- Ileocaecal valve dysfunction
- Post-infectious autoimmunity: anti-CdtB antibodies cross-reacting with vinculin via molecular mimicry
5. IMO: Intestinal Methanogen Overgrowth
IMO (Intestinal Methanogen Overgrowth) is a distinct entity caused by archaea — primarily Methanobrevibacter smithii — not bacteria. These methanogens consume hydrogen produced by bacteria and generate methane.
Key insight: Methane is not an inert gas. It acts as an active paralytic agent on intestinal smooth muscle, causing severe refractory constipation — frequently misdiagnosed as IBS-C (constipation-predominant irritable bowel syndrome).
M. smithii can colonise the entire gastrointestinal tract and is notoriously difficult to eradicate. Historically, treatment requires dual-antibiotic protocols (rifaximin + neomycin or metronidazole).
6. Berberine's Multi-Target Mechanisms in SIBO Eradication
Berberine deploys a unique multi-target therapeutic strategy that goes beyond simple antimicrobial activity:
6.1 Direct antimicrobial and anti-biofilm activity
Berberine inhibits FtsZ protein assembly (prokaryotic tubulin homologue), which arrests bacterial cell division. It also reduces fimbrial gene expression, stripping bacteria of their adhesion and biofilm formation capacity — a major resistance mechanism in chronic SIBO.
6.2 Microbiome restructuring
Berberine is not a blind antimicrobial. It exerts selective microbiome restructuring:
| Species/Phyla decreased | Species/Phyla increased |
|---|---|
| Desulfovibrio | Bacteroidetes |
| Streptococcaceae | Clostridia |
| Proteus | Lactobacillales |
| Saccharibacteria | Ruminococcus |
| Deferribacteres | Bifidobacterium |
| Actinobacteria | Lactobacillus |
| Coprococcus (key biomarker) | Akkermansia muciniphila |
Key insight: Coprococcus suppression is the primary biomarker of berberine efficacy (0.18 ± 0.13% vs 1.09 ± 0.20%, P<0.001). The simultaneous enrichment of Akkermansia muciniphila — a key species for gut barrier health — suggests deep, beneficial ecosystem remodelling.
6.3 Tight junction restoration
Berberine stimulates tight junction protein expression (occludin, claudins, ZO-1), repairing the intestinal epithelial barrier. This mechanism halts bacterial and endotoxin translocation to the systemic circulation, reducing the chronic low-grade inflammation associated with SIBO.
6.4 The motility paradox: opioid receptor agonism
Berberine is an agonist of mu-opioid (MOR) and delta-opioid (DOR) receptors, which slows intestinal transit — a classically antidiarrhoeal effect. This is reversed by naloxone, β-funaltrexamine (mu-selective) and naltrindole (delta-selective), but not by nor-binaltorphimine (kappa-selective).
Paradox resolution: Slowing transit via opioid agonism prolongs contact time between berberine and pathogenic bacteria, enabling more complete eradication. Once the overgrowth is decimated, symptomatic relief far outweighs the mild motility delay. This mechanism also explains berberine's long-term superiority over rifaximin.
7. Clinical Trials: From Chedid 2014 to BRIEF-SIBO 2024
7.1 Chedid et al. 2014 (Johns Hopkins)
This pioneering study compared botanical therapy (including berberine sulfate 400 mg) with rifaximin 1200 mg/day. Result: statistically equivalent SIBO eradication. Key finding: berberine-containing botanical formulae proved effective as rescue therapy for rifaximin non-responders.
7.2 BRIEF-SIBO 2024 (Peking University Third Hospital)
This randomised controlled trial (ChiCTR2200057554) enrolled 186 patients diagnosed by lactulose breath test (LHMBT). Protocol: berberine HCl 400 mg twice daily vs rifaximin 400 mg twice daily, for 2 weeks with 6-week follow-up. Non-inferiority design (α=0.025, power=0.8).
| Endpoint | Berberine | Rifaximin | Statistic |
|---|---|---|---|
| Week 2 eradication (ITT) | 45% | 51% | Z=2.407, P=0.008 — non-inferior |
| Week 6 eradication (ITT) | 53% | 44% | Z=4.324, P<0.001 — SUPERIOR |
| Week 6 eradication (PP) | 54% | 45% | Z=4.077, P<0.001 |
Key insight: Rifaximin shows rapid bacterial relapse visible on breath test curves between weeks 2 and 6. Berberine maintains a stable negative breath test, creating a durably inhospitable environment for bacterial overgrowth. Symptomatically: rifaximin primarily improves bowel frequency and consistency, while berberine resolves severe distension and bloating at Week 6.
8. Network Meta-Analysis and SUCRA Rankings
A network meta-analysis spanning 30 randomised controlled trials (n=1,552, 12 interventions) established the following SUCRA (Surface Under the Cumulative Ranking) rankings for SIBO eradication:
| Rank | Intervention | SUCRA |
|---|---|---|
| 1 | Berberine monotherapy | 82.5% |
| 2 | Botanical/antibiotic combination | 72.5% |
| 3 | Rifaximin + prokinetic | 66.9% |
| 4 | Quinolones | 64.0% |
| 5 | Aminoglycosides | 59.4% |
| 6 | Rifaximin monotherapy | 57.5% |
| 7 | Probiotics | 55.3% |
| 8 | Prokinetic monotherapy | 53.5% |
| 9 | Metronidazole | 40.2% |
| 10 | Ursodeoxycholic acid | 25.9% |
Key insight: Berberine monotherapy achieves the highest SUCRA score of all evaluated interventions — 25 points above rifaximin alone. This is all the more remarkable given that berberine does not require a medical prescription and has a favourable safety profile.
9. Specific Efficacy in IMO
Subgroup analysis from the BRIEF-SIBO trial reveals particularly encouraging results for IMO (methane-SIBO):
- Breath test normalisation in CH4-SIBO: 44.7% (vs 33.9% in H2-SIBO)
- Clinical symptom remission in CH4-SIBO: 73.2%
Key insight: Berberine halts methane production by archaea, removing the paralytic brake on intestinal smooth muscle and resolving constipation — despite its opioid agonist properties. This result is clinically transformative for IMO patients who often fail conventional dual-antibiotic protocols. Facial erythema resolution is frequently observed by week 10.
10. Safety, Contraindications and Drug Interactions
10.1 Dosage and tolerance
Berberine is considered safe at standard doses of 1.0–1.5 g/day in divided doses, for durations up to 6 months. Gastrointestinal side effects are common but transient and dose-dependent: nausea, cramping, bloating, gas, diarrhoea or constipation.
10.2 Absolute contraindications
- Pregnancy and breastfeeding: absolute contraindication
- Neonates: risk of kernicterus through bilirubin conjugation inhibition
10.3 Major drug interactions
| Drug class | Risk | Mechanism |
|---|---|---|
| Antidiabetics | Hypoglycaemia | Synergistic effect on blood glucose |
| Antihypertensives | Hypotension | Potentiation of vascular effect |
| Immunosuppressants | Graft rejection | Immune modulation |
Clinical recommendation
Berberine use should always be supervised by a qualified healthcare professional, particularly in patients on chronic medication. Self-supplementation without professional guidance carries significant interaction risks.
11. Our Personalised Approach at Diaeta
The data presented in this review confirm berberine's exceptional therapeutic potential in SIBO and IMO. However, no molecule, however promising, replaces a comprehensive, individualised therapeutic strategy.
What we promise you
- Never hungry: every nutrition plan accompanying SIBO treatment is designed to be satisfying and delicious, even during dietary modulation phases
- No unnecessary eliminations: we precisely identify individual triggers rather than imposing generic protocols
- Evidence-based guidance: every recommendation is grounded in the most current scientific literature, including BRIEF-SIBO data and network meta-analyses
- Personalised strategies: your treatment plan is adapted to YOUR breath test profile, YOUR microbiome and YOUR goals
How we support you
- Comprehensive assessment: analysis of your breath test results, medical history, current medication and symptom profile
- Multi-modal eradication strategy: personalised integration of berberine, nutritional support and microbiome management
- Guided reintroduction phase: progressive, structured dietary re-expansion after eradication
- Relapse prevention: prokinetic protocols, MMC optimisation and long-term monitoring
Observed outcomes
With our personalised approach, our patients report:
- Significant reduction in bloating and distension through multi-target strategies
- Lasting improvement in digestive comfort without early relapse
- Restoration of eating enjoyment as symptoms diminish and the dietary repertoire expands
Ready to transform your digestive comfort?
SIBO, IMO, chronic bloating or refractory constipation? Our evidence-based, personalised approach supports you towards lasting eradication and a serene digestive life — without unnecessary deprivation.
Scientific References
- Chedid V, Dhalla S, Clarke JO, et al. Herbal therapy is equivalent to rifaximin for the treatment of small intestinal bacterial overgrowth. Glob Adv Health Med. 2014;3(3):16-24.
- Zhang Y, Gu Y, Ren H, et al. Berberine versus rifaximin for SIBO eradication: the BRIEF-SIBO randomised controlled trial. Gut. 2024;73(7):1105-1115.
- Habtemariam S. Berberine pharmacology and the gut microbiota: a hidden therapeutic link. Pharmacol Res. 2020;155:104722.
- Imenshahidi M, Hosseinzadeh H. Berberine and barberry (Berberis vulgaris): a clinical review. Phytother Res. 2019;33(3):504-523.
- Dominguez-Bello MG, Godoy-Vitorino F, Knight R, Blaser MJ. Role of the microbiome in human development. Gut. 2019;68(6):1108-1114.
- Pimentel M, Saad RJ, Long MD, Rao SSC. ACG Clinical Guideline: Small Intestinal Bacterial Overgrowth. Am J Gastroenterol. 2020;115(2):165-178.
- Rezaie A, Buresi M, Lembo A, et al. Hydrogen and methane-based breath testing in gastrointestinal disorders: the North American Consensus. Am J Gastroenterol. 2017;112(5):775-784.
- Feng X, Wang K, Cao S, Ding L, Qiu F. Pharmacokinetics and excretion of berberine and its nine metabolites in rats. Front Pharmacol. 2021;12:579591.
- Cicero AFG, Baggioni A. Berberine and its role in chronic disease. Adv Exp Med Biol. 2016;928:27-45.
- Takahara M, Takaki A, Hiraoka S, et al. Berberine improved experimental chronic colitis by regulating interferon-gamma and IL-17A-producing lamina propria CD4+ T cells through AMPK activation. Sci Rep. 2019;9(1):11934.
- Chen C, Yu Z, Li Y, et al. Effects of berberine in the gastrointestinal tract — a review of actions and therapeutic implications. Am J Chin Med. 2014;42(5):1053-1070.
- Lauritano EC, Gabrielli M, Scarpellini E, et al. Small intestinal bacterial overgrowth recurrence after antibiotic therapy. Am J Gastroenterol. 2008;103(8):2031-2035.